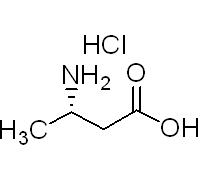
L-beta-Homoalanine hydrochloride
(S)-3-aminobutanoic acid hydrochloride
CAS: 58610-41-6
Molecular Formula: C4H10ClNO2
L-beta-Homoalanine hydrochloride - Names and Identifiers
L-beta-Homoalanine hydrochloride - Physico-chemical Properties
| Molecular Formula | C4H10ClNO2 |
| Molar Mass | 139.58 |
| Boling Point | 223.6°C at 760 mmHg |
| Flash Point | 89°C |
| Vapor Presure | 0.0352mmHg at 25°C |
| BRN | 4754597 |
| Storage Condition | Inert atmosphere,Room Temperature |
| MDL | MFCD01862873 |
L-beta-Homoalanine hydrochloride - Risk and Safety
| WGK Germany | 3 |
L-beta-Homoalanine hydrochloride - Reference Information
| Uses | L-bet-high alanine hydrochloride is an amino acid derivative. There are reports that it can be used to prepare compounds for the treatment of HBV infection. |
| Preparation | In a 5mL round bottom flask with a glass stopper, heat (E)-butyl-2-enoic acid ethyl ester (20 mmol, 2.28g) and benzylamine (44 mmol, 481 ml) at 60°C. The reaction mixture was stirred for 30 hours. The completion of the thermal Michael addition reaction was monitored by 1H-NMR spectroscopy (5 μl sample)(93.0% total conversion, 91.0% product-related conversion). Lipase (50 mgmmol-1,100mg) from Candida antarctica B(CAL-B) was added to the reaction mixture. The reaction mixture was stirred for a further 16 hours at 60°C. The completion of the reaction was monitored by 1H-NMR spectroscopy (61.2% conversion). The reaction mixture was separated from the immobilized enzyme by filtration. The reaction mixture was washed with methyl tert-butyl ether (MTBE,20mL). The resulting organic solution was washed three times with a saturated NaHCO3(1mL) aqueous solution to give 3-aminobutanoic acid and N-benzyl (R)-3-(benzylamino) butanamide. An aqueous solution of NaOH(1M,14.4mL) was added to the organic layer containing (S)-3-aminobuty-2-enoic acid in a 10mL round bottom flask. The resulting reaction mixture was stirred at room temperature for 16 hours. The reaction course was monitored according to the consumption of TLC (ethyl acetate: 2-PrOH,95:5(v/v),0.1% diethylamine (S)-3-aminobutyl-2-enoic acid). After the reaction is completed, the water layer is separated by a separatory funnel. The organic phase was extracted 3 times with NaOH aqueous solution (0.1M,1mL) to obtain the intermediate. An ion exchanger Merck-III(14.4g) was added to the aqueous layer of the resulting intermediate. Stir the resulting mixture for one hour. Wash the mixture with water (40mL). The compound was eluted with HCl aqueous solution (1M,70mL). Hydrochloric acid aqueous solution was evaporated under reduced pressure at 60 ℃ to obtain L-bet-hyperalanine hydrochloride. |
Last Update:2024-04-09 19:05:04
Supplier List
Spot supply
Product Name: L-β-Homoalanine hydrochloride Visit Supplier Webpage Request for quotationCAS: 58610-41-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (S)-3-aminobutanoic acid hydrochloride Request for quotation
CAS: 58610-41-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 58610-41-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: L-beta-Homoalanine hydrochloride Request for quotation
CAS: 58610-41-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 58610-41-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: L-β-Homoalanine hydrochloride Visit Supplier Webpage Request for quotation
CAS: 58610-41-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 58610-41-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: L-β-Homoalanine hydrochloride Visit Supplier Webpage Request for quotationCAS: 58610-41-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (S)-3-aminobutanoic acid hydrochloride Request for quotation
CAS: 58610-41-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 58610-41-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: L-beta-Homoalanine hydrochloride Request for quotation
CAS: 58610-41-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 58610-41-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: L-β-Homoalanine hydrochloride Visit Supplier Webpage Request for quotation
CAS: 58610-41-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 58610-41-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



